-
Welcome, Guest
- ( My Account | View Cart | Login )
Shopping Cart Items: 0
Sub-Total :
US$0.00
|
|
| Our Local Time Is 10:37:05 PM. |
| Call us at 818-786-0600. We are here to help! |
 |
Need a New Laboratory Water System? We have many to choose from. We can save you thousands on Complete Systems and replacement filters for most brands. |
| Laboratory Water Systems | Laboratory Process Filters | Laboratory Equipment | Water Treatment |
* Flat Rate UPS shipping when able to ship via UPS and is in the USA excluding Hawaii and Alaska. Larger Items may not be able to ship via UPS, in that case freight charges will be quoted seperately. International shipping will be quoted after the order is placed. You will have the opportunity to cancel before we finalize your order. Terms and conditions Credit Application Privacy Policy Google Apps List All Products |

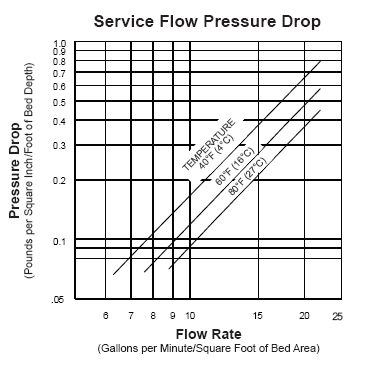 .
. 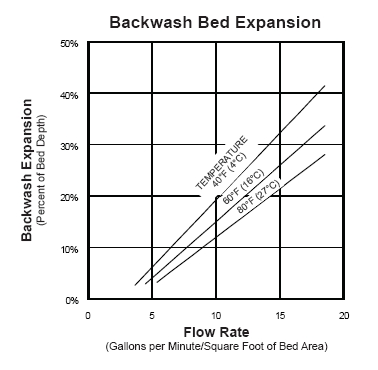 ..
..  ...
...











